Properties of soaps and detergents
Soap – Characteristics And Uses
Soaps are excellent cleansing agents and have good biodegradability. A serious drawback which reduces their general use, is the tendency for the carboxylate ion to react with Ca+ and Mg+ ions in hard water. The result is a water insoluble salt which can be deposited on clothes and other surfaces. These hard water plaques whiten fabric colors and also create rings found in sinks and bath tubs. Another problem with using soaps is their ineffectiveness under acidic conditions. In these cases, soap salts do not dissociate into their component ions, and this renders them ineffective as cleansing agents.
Although primarily used for their cleansing ability, soaps are also effective as mild antiseptics and ingestible antidotes for mineral acid or heavy metal poisoning. Special metallic soaps, made from soap and heavier metals, are used as additives in polishes, inks, paints, and lubricating oils.
Detergent Physical Characteristics
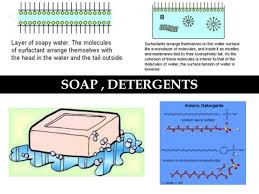
The concentration at which micelles begin to form is the critical micelle concentration (CMC). The CMC is the maximum monomer concentration and constitutes a measure of the free energy of micelle formation. The lower the CMC, the more stable the micelle and the more slowly molecules are incorporated into or removed from the micelle. The structure of the hydrophobic region of the detergent can affect the micelle structure. An increase in the length of the hydrophobic hydrocarbon chain of ionic detergents results in an increased micelle size and a lower CMC, as fewer molecules are needed to construct a micelle.
The average number of monomers in a micelle is the aggregation number. The CMC and aggregation number values are highly dependent on factors such as temperature, pH, ionic strength, and detergent homogeneity and purity. Slight discrepancies in reported values for CMC and aggregation number may be the result of variations in the analytical methods used to determine the values. Aggregation number values are also shifted by concentration, since the number of detergent molecules per micelle may increase if the concentration is above the CMC.
Ease of removal or exchange is an important factor in the selection of a detergent. Some of the more common detergent removal methods include:
- Dialysis
- Gel filtration chromatography
- Hydrophobic adsorption chromatography
- Protein precipitation
The CMC value associated with the detergent is a useful guide to hydrophobic binding strength. Detergents with higher CMC values have weaker binding and are subsequently easier to remove by dialysis or displacement methods. Detergents with low CMC values require less detergent in order to form micelles and solubilize proteins or lipids.
Another useful parameter when evaluating detergents for downstream removal is the micelle molecular weight, which indicates relative micelle size. Smaller micelles are more easily removed and are usually desirable when protein-detergent complexes are to be separated based on the molecular size of the protein. The micelle molecular weight may be calculated by multiplying the aggregation number by the monomer molecular weight.
The cloud point is the temperature at which the detergent solution near or above its CMC separates into two phases. The micelles aggregate, typically forming a cloudy phase with high detergent concentration, while the balance of the solution becomes detergent-depleted. The resulting two-phase solution can be separated, with the extracted protein being located in the detergent-rich phase. Detergents with low cloud point temperatures, such as TRITON® X-114 (cloud point ~23 °C) are recommended for use with proteins since high cloud point temperatures may denature solubilized proteins. The cloud point can be affected by changes in detergent concentration, temperature, and the addition of salt or polymers such as dextran and polyethylene glycol. Note that the detergent-rich phase is also contingent on the specific detergent(s) and salt concentration; under some conditions the phase may be clear rather than cloudy and be located as either the upper or lower phase of the solution. In non-ionic detergents, this behavior has been applied in the phase separation and purification of membrane proteins.2
Detergent Types and Selection
When selecting a detergent, the first consideration is usually the form of the hydrophilic group:
- Anionic
- Cationic
- Non-ionic
- Zwitterionic (ampholytic)
Anionic and cationic detergents are considered biologically “harsh” detergents because they typically modify protein structure to a greater extent than neutrally charged detergents. The degree of denaturation varies with the individual protein and the particular detergent and concentration. Ionic detergents are more sensitive to pH, ionic strength, and the nature of the counter ion, and can interfere with downstream charge-based analytical methods.
Non-ionic detergents are considered to be “mild” detergents because they are less likely than ionic detergents to denature proteins. By not separating protein-protein bonds, non-ionic detergents allow the protein to retain its native structure and functionality, although detergents with shorter hydrophobic chain lengths are more likely to cause protein deactivation. Many nonionic detergents can be classified into three structure types:
- Poly(oxyethylene) ethers and related polymers
- Bile salts
- Glycosidic detergents
Poly(oxyethylene) ethers and related detergents have a neutral, polar head and hydrophobic tails that are oxyethylene polymers (e.g. Brij® and TWEEN®) or ethyleneglycoether polymers (e.g. TRITON®). The tert-octylphenol poly(ethyleneglycoether) series of detergents, which includes TRITON X-100 and IGEPAL® CA-630, have an aromatic head that interferes with downstream UV analysis techniques.
Bile salts have a steroid core structure with a polar and apolar orientation, rather than the more obvious nonpolar tail structure of other detergents. Bile salts may be less denaturing than linear chain detergents with the same polar head group.
Glycosidic detergents have a carbohydrate, typically glucose or maltose, as the polar head and an alkyl chain length of 7-14 carbons as the polar tail.
Zwitterionic detergents have characteristics of both ionic and non-ionic detergent types. Zwitterionic detergents are less denaturing than ionic detergents and have a net neutral charge, similar to non-ionic detergents. They are more efficient than non-ionic detergents at disrupting protein-protein bonds and reducing aggregation. These properties have been used for chromatography, mass spectrometry, and electrophoresis methods, and solubilization of organelles and inclusion bodies.
Non-detergent sulfobetaines (NDSB), although not detergents, possess hydrophilic groups similar to those of zwitterionic detergents but with shorter hydrophobic chains. Sulfobetaines do not form micelles. They have been reported to improve the yield of membrane proteins when used with detergents and prevent aggregation of denatured proteins.
Uses for Detergent
Powder and liquid detergent can be used for other purposes besides cleaning clothes or dishes. This can save you money from having to buy multiple cleaning products.
All-Purpose Cleaner
Either form of detergent can be used to clean tiles, floors, counters, tubs and toilets. Mix 3/4 of a cup of bleach, 1 cup of detergent and 1 gallon of hot water together and pour it into spray bottles for a supply of all-purpose cleaner.
Moss Killer
Sprinkle powdered detergent on moss that is growing in the cracks of your steps, sidewalk or driveway. Give it a few days to turn brown, then brush it from the cracks with a broom.
Oil Spills
Powdered detergent can absorb oil that’s spilled on a garage floor or on the street.
Carpet Cleaning
Both types of cleaner can be added to carpet steam cleaners to make the carpet smell fresher and boost the appliance’s cleaning power.
Drains
Instead of buying Drano to clean out a drain, put 1/4 cup of liquid detergent into the drain, then pour in a boiling pot of water after a minute to flush out to blockage.
Bubbles
If kids like to make bubbles with wants or play with bubble-making guns, one can make the bubble solution oneself by mixing liquid detergent with water.